Research Ethics Committees
There are two levels of Research Ethics Committees in the Institute, at Faculty/Department level and at Institute level (IREC).
All staff and students of ATU Sligo are required to plan and conduct their research investigations in accordance with appropriate ethical standards. Staff should ensure that they have knowledge of any relevant disciplinary guidelines on research ethics and that any empirical research has the required approval by the Research Ethics Committees.
All potential applicants should be familiar with the Research Ethics Policy and the related Research Ethics Procedure.
The function of each of the Research Ethics Committees is to safeguard the health, welfare and rights of human participants and researchers in research studies. For any research proposal to gain ethical approval it must be necessary and of a design that minimises predictable risk to both the research participant and the researcher.
The Research Ethics Committees aspires to provide comprehensive and independent reviews of the ethics of proposed studies, acting in accordance with good ethical practice as dictated by relevant EU Directives, National legislation and practice guidelines. If the proposed research involves patients (i.e. people who are receiving treatment as a result of an illness) the applicant should seek confirmation of the need to submit their application to the relevant hospital’s Research Ethics Committees.
Before Applying
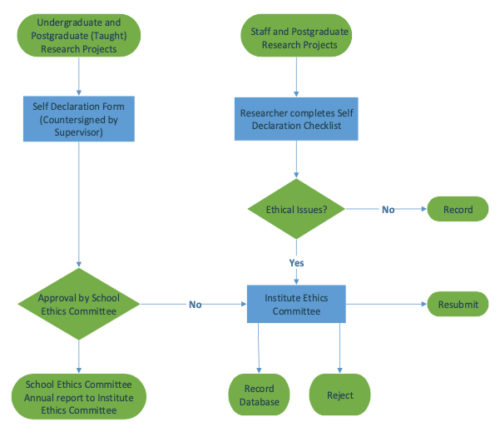
Applicants should consider which research ethics committee that their application needs to be considered at by reviewing the flowchart below.
The IREC only reviews applications from staff research projects and post graduate research students and those applications which the Schools Research Ethics Committees feel require further consideration by the IREC.
The IREC does NOT consider applications from taught Masters Students or undergraduate students.
How to Apply?
Applications to Faculty/Department Research Ethics Committee:
Please liaise with your lecturer/ supervisor to ascertain the process for applying to your School/ Department research ethics committee.
Applications to Institute Research Ethics Committee:
Applicants are requested to complete a Research Ethics Approval Application Form.
Please email your application to both chris.omalley@nullatu.ie and emma.flanagan@nullatu.ie.
We will acknowledge receipt of your application by providing a Reference number to you by email.
The IREC recommends to view the videos below before making an application.
Where the research involves interviewing or surveying people, the application also needs to be accompanied by:
- A copy of the Consent Form and
- A copy of the Participant Information Sheet (advising participants of what the research is for, how their role in it will work and what may/is to be done with the results) of the research and
- A copy of the Questionnaire or Interview Guidelines to be used
- Together with the applicant’s signature (Download Signature Page Template).
These documents should be emailed to the Chair of the Institute’s Research Ethics Committee at chris.omalley@nullatu.ie. and emma.flanagan@nullatu.ie.
In advance of the deadline for applications for the Committee meeting where you wish the application to be considered.
Please note the applications will not be processed without the signature page. Signatures must be original.
In cases where other research ethics committees have reviewed the application, copies of their responses must be uploaded as part of the application process.
As soon as feasible after the closing date for receipt of applications, the application will be allocated an IREC reference number. This number will be sent to the principal investigator to acknowledge receipt of the application. This number should be quoted on any further correspondence between the IREC and the principal investigator.
IREC Committee Meeting & Submission dates September 24 - May 2025
Feedback will be provided by the committee in a constructive capacity that is intended to support the applicants in their research.

National and international guidance documents
- International Ethical Guidelines for epidemiological studies WHO
- Operational_Procedures1 for reserach ethics committees
- The Irish council for Bioethics 2004
- HRB policy of research ethics
- Ethics for researchers EUROPE
- inf206 guidelines for internet mediated research
Animal / Genetic research
- HRB_Paper_on_the_Legal_and_Ethical_Considerations_for_Genetic_Research
- HRB_Policy_on_use_of_animals_in_research
Data Protection
- Webinar-on-GDPR-and-health-research-regulations-2018
- Data protection guidelines on reserach in the health sector
- Data Protection Act 2003